Isomerization
isomerization
[ī‚säm·ə·rə′zā·shən](chemistry)
A process whereby a compound is changed into an isomer; for example, conversion of butane into isobutane.
McGraw-Hill Dictionary of Scientific & Technical Terms, 6E, Copyright © 2003 by The McGraw-Hill Companies, Inc.
The following article is from The Great Soviet Encyclopedia (1979). It might be outdated or ideologically biased.
Isomerization
the conversion of a chemical compound into its isomer. In isomerization the carbon skeleton of a molecule can change as well as the nature of the functional groups and their positions, ring contraction or enlargement can occur, and so forth. Thus treatment of straight chain saturated (aliphatic) hydrocarbons with aluminum chloride (A1C13) yields branched chain hydrocarbons (for example, butane to isobutane):
CH3CH2CH2CH3 → (CH3)2CH—CH3
These conversions occur when petroleum is refined (in cracking, pyrolysis, and reforming processes) and in part result in the formation of high-octane gasolines. Cyclohexanone oxime is iso-merized by acid to caprolactam, the starting material in the production of a nylon synthetic fiber (kapron):
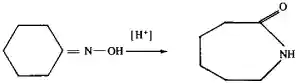
This isomerization is a particular case of the Beckmann rearrangement. Among the other isomerization processes of practical importance are the conversion of ethylene oxide to acetaldehyde; the isomerization of o- and m-xylene to p-xylene, from which terephthalic acid is prepared by oxidation; and the conversion of hydrazobenzene into benzidine. The concept of isomerization also includes the interconversion of geometric isomers, for example, maleic (I) acid and fumaric (II) acid:
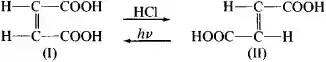
as well as numerous cases of racemization.
B. L. DIATKIN
The Great Soviet Encyclopedia, 3rd Edition (1970-1979). © 2010 The Gale Group, Inc. All rights reserved.